What is FH?
Overview
Hypercholesterolemia is an excess of cholesterol in the bloodstream that increases the risk of heart disease and stroke. Familial hypercholesterolemia is when the excess of cholesterol is caused by an inherited genetic condition. It is an autosomal dominant genetic disease, which means that women and men are affected equally and that siblings and children of a person with FH have a 50% risk of getting the defect. The condition may be discovered if you have a routine health check. People with FH have high cholesterol levels from birth and this leads to an early development of atheroma (fatty lumps within the arteries) and associated cardiovascular diseases (angina, heart attack, stroke, transient ischaemic attack and peripheral vascular diseases).
Causes of FH
Familial hypercholesterolemia is caused by a defect in a few genes responsible for the way cholesterol is handled in the body. The cholesterol is transported in the bloodstream within specific transporters called low-density lipoproteins (LDL), also referred to as “bad cholesterol”. People with familial hypercholesterolemia have high levels of LDL-cholesterol as their body cannot effectively remove the LDL particles from their bloodstream, which causes the development of atheroma and premature cardiovascular diseases. Generally, the defective gene is inherited from one parent (heterozygous inheritance), and affects about 1 in 250 people. Some patients can inherit the defective gene from both parents (homozygous inheritance), which will result in a rare but more severe condition (with a chance of 1 in 250,000).
Symptoms
Patients with familial Hypercholesterolemia may feel no symptoms of the disease, and their first presentation may be an acute myocardial infarction (heart attack).
Clinical Findings
Possible signs of familial hypercholesterolemia include:
- High levels of total cholesterol and LDL cholesterol, dating back to childhood.
- Development of cardiovascular disease at a young age, in some people as young as 20 years old.
- A strong family history of high levels of total cholesterol and LDL cholesterol and/or early heart attack (“early” means before 55 y for men and before 65 y for women).
- Elevated and therapy-resistant levels of LDL in either or both parents.
- Presence of xanthomas –fatty cholesterol-rich deposits in the skin– usually found around the elbows, knees, buttocks, knuckles and tendons.
- Xanthelasmas – cholesterol deposits in the eyelids.
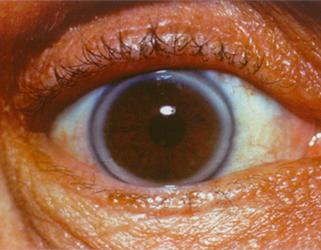
- Premature Corneal Arcus – a white ring around the cornea of the eye, before the age of 45 (this could be normal after the age of 45).
-
Tendinous xanthomas in an untreated adult patient with heterozygous familial hypercholesterolemia Courtesy of Dr. Jacques Genest
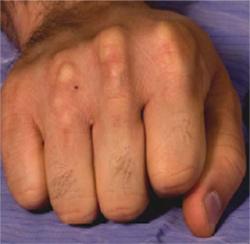
-
Normal (left) versus xanthomatous Achilles tendons (right) and their radiological assessment Courtesy of Dr. Jean Davignon and Dr. Robert Dufour
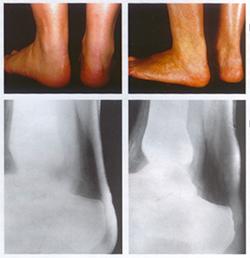
-
Xanthomas in the knees in a patient with heterozygous familial hypercholesterolemia Courtesy of Dr. Jean Davignon and Dr. Robert Dufour
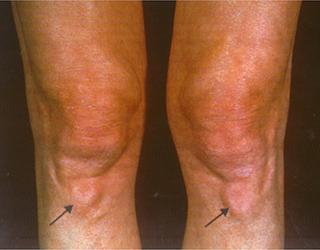
-
Xanthelasmas around the eyes in an untreated adult patient with heterozygous familial hypercholesterolemia Courtesy of Dr. Jacques Genest
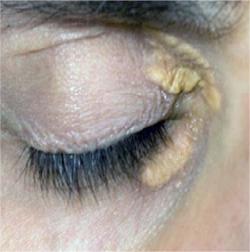
-
Corneal arcus in a patient with heterozygous familial hypercholesterolemia Courtesy of Dr. Jean Davignon and Dr. Robert Dufour
Diagnosis
Familial hypercholesterolemia is a disease that is both underdiagnosed and undertreated. The aim of the FH Canada registry is to improve the detection and management of individuals and families with FH in Canada. The project will identify those affected by FH with the aim of lowering their high cholesterol and thus preventing early cardiovascular disease such as heart attack and stroke.
You may have been diagnosed by chance if you had a health screening check-ups or noticeable fatty deposits on the skin or around the eyes, or you may have been advised to have yourself checked when a member of your family was diagnosed with familial hypercholesterolemia or had a heart attack before the age of 55.
The diagnosis of familial hypercholesterolemia is based on physical examination and laboratory testing. A physical exam will be conducted by your physician and will help identify any types of fatty deposits or lesions that have developed as a result of the elevated cholesterol levels (xanthomas, xanthelasmas and corneal arcus). Your physician will also ask about your personal medical history, as well as your family medical history. Laboratory testing mainly includes a blood test for cholesterol levels. Familial hypercholesterolemia is diagnosed in the presence of increased cholesterol levels in the absence of secondary causes of hypercholesterolemia (severe or untreated hypothyroidism, nephrotic syndrome [a disease of the kidneys], liver disease [biliary cirrhosis], some drugs such as antiretroviral agents). In adults, familial hypercholesterolemia is suspected if your blood total cholesterol level is 7.5 mmol/L or greater, or if your LDL-cholesterol levels are 4.9 mmol/L or greater. In children, the cut-point levels are 6.7 mmol/L for total cholesterol and 4.0 mmol/L for LDL-cholesterol. In some clinics, studies of heart function (stress test) and genetic testing may be included.
A genetic testing for familial hypercholesterolemia is now also available. If you know there is a genetic defect causing familial hypercholesterolemia in your family, your DNA can be tested. In fact, if you have FH, each of your close family members has a 50% chance of also having FH, including parents, siblings, children. This means that if you are diagnosed with FH, your whole family should get tested. If your cholesterol is high and you have a family history of high cholesterol or premature heart disease, familial hypercholesterolemia is suspected, and you should have access to a genetic testing. Please ask you family doctor on how to proceed.
Treatment
The overall goal of treatment is to lower the risk for atherosclerotic heart disease by lowering the LDL cholesterol levels in the blood stream.
Lifestyle:
Being a genetic disorder, familial hypercholesterolemia is not caused by an unhealthy lifestyle. However, keeping yourself in the best physical condition will help prevent future problems.
A healthy lifestyle includes:
- Eating healthily. You will almost certainly be referred to a dietitian to help you make appropriate changes in your eating habits.
- Exercise.
- Avoiding smoking.
- Maintaining a normal weight.
- Stress management
Medication:
You will be offered medications to help bring your cholesterol level down. The first and more effective choice are drugs called “statins” (HMG-CoA-reductase inhibitors). The most common statins used in Canada are atorvastatin (Lipitor), rosuvastatin (Crestor) and simvastatin (Zocor). If the level of cholesterol does not come down, another medicine called ezetimibe (Ezetrol), which decreases cholesterol absorption in the small intestine, is sometimes added. Bile acid binding resins, nicotinic acid (niacin), gemfibrozil, fenofibrate and estrogen replacement therapy in postmenopausal women can also be prescribed. The third-line treatment for familial hypercholesterolemia is the use of monoclonal antibody inhibitors of PCSK9, especially if your LDL-cholesterol levels have not been reduced enough by statin and ezetimibe therapy. Some patients with extreme levels of cholesterol, such as the homozygous patients, may need apheresis, a treatment which filters LDL cholesterol out of the blood. These patients should be followed by specialized lipid clinics.
Your general practitioner or family doctor may want to discuss referring you to a specialist. A specialist can advise you about your treatment and arrange the testing of close members of your family. You may also refer to the list of FH Canada Registry participating clinics.
The prognosis for people with familial hypercholesterolemia is excellent if you maintain a healthy lifestyle, have regular check-ups and take your medication without fail. The most significant complications are heart disease or another cardiovascular disease that may develop at a younger age than usual.
Maintaining a good lifestyle is the cornerstone of preventive strategies in children, and all children with a presumptive diagnosis of FH should undergo 6 to 12 months of extensive lifestyle changes including diet, exercise and a tobacco-free environment. As in adults, if drug treatment is required, statins should be used as first-line therapy with ezetimibe as a second-line agent. Bile acid binding resins should be considered third-line therapy.
FH Canada Registry Information
Purpose of the FH Canada Initiative
The aim of the FH Canada registry is to improve the detection and management of individuals and families with FH in Canada. The project will identify those affected by FH with the aim of lowering their high blood cholesterol and thus preventing early cardiovascular disease such as heart attack and stroke. Patients with other severe lipid disorder will also be included.
Individuals with FH have a very high risk of developing early cardiovascular diseases. Early treatment is useful to prevent early heart disease in people with FH. The purpose of the project is to identify and help get treatment started for people who are unaware that they have FH or are not being treated properly. In Canada, the burden of disease is estimated to be approximately 140,000 patients. Our goal is to create a list of individuals (a registry) with the diagnosis of FH in Canada. This will greatly simplify education and treatment of these people in the future.
Strategy of the study
We use the method of “cascade screening”, which involves testing of lipid levels in all primary (parents, siblings, and children), secondary (aunts, uncles, grandparents, grandchildren) and third-degree (cousins, nephews, nieces) relatives of the family member diagnosed with FH. This method has been used in other countries with a great deal of success and it has been established that this process of “seek and treat” saves both lives and money through this health promotion/disease prevention strategy. The data collected includes personal data, contact information for you and your family physician, medical and familial history, lifestyle data, and laboratory findings. This data will be used only for the purpose of monitoring your treatment and your health. Only the study doctor will have access to this data, it will be kept confidential. The personal data will not be shared with other study doctors or linked with other databases. You will not be identified and will be assign a randomly generated number, which cannot be trace back to your medical chart. The data collected in the national database will allow each province to determine the number of FH cases in the province and to determine health outcomes, resource utilization and health economics, essential information in order to provide better healthcare. It will also serve for Canada-wide statistics on FH; no identifier (name, ID number, date of birth, address) will be included.
Who can participate?
You may participate in this study if you have a:
- Diagnostic criteria for FH, which are:
- Family and/or personal history of high cholesterol.
- Family and/or personal history of heart disease.
- Abnormal growth on tendons, accumulation of fatty material in the eye.
- Family history of FH.
- Severe disorder of cholesterol and other lipids in the blood.
What are the possible benefits to participating – why should you join?
- Early diagnosis of FH, particularly of family members who may not yet know they have FH (cascade screening).
- Expert care and better management of high cholesterol levels resulting in the prevention of heart disease in patients with FH.
- Support and information for individuals with FH.
- New information obtained from the registry will be used to guide and inform research studies directed at improving our knowledge and treatment of FH, which may benefit patients with FH or other persons with elevated blood lipids in the future.
- One of the goals of the registry is to develop comparative statistics on the patients in the registry so that patients can see how they are doing in relation to other FH patients. We hope that this will encourage patients to become involved in their own health care.
- Access to on-going trials and to novel therapies (new drugs).
What does the study involve?
You will be asked about your family history for high cholesterol and your personal medical history related to diseases of the heart or the blood vessels and use of medications (this first visit should take about 30 minutes). If you are already on lipid-lowering medications, we will ask you for information about your pre-treatment and on-treatment cholesterol levels and provide you with information on achieving proper cholesterol levels using Canadian guidelines for the diagnosis and treatment of high cholesterol. If you have never been tested, we will give you a requisition to get your blood drawn and then we will follow up with you when we get the results. The lab will take about 15 ml (1.5 tablespoons) of blood. With your approval, a sample of blood plasma and DNA will be kept at the participating lipid clinic. If you have been tested and are uncertain if you do or do not have FH, we will discuss the test results with you or -with your approval- with your family physician. With your approval, your family physician will be notified of your participation in the study so that your study doctor and your family doctor can provide proper medical care. All your study data will be available to you and, if you wish, also sent to your family physician. With your approval, you will be followed up every year about your management. You will be interviewed about your health, current treatment and laboratory findings. This can be done by phone interview or if you wish, by personal visit. There are no costs to you for taking part in the FH Canada registry, and you will not be paid for taking part either.
Consent
You can participate in the FH Canada registry if you are a patient with FH or a family member of a patient diagnosed with FH, or because you present a severe disorder of cholesterol and other lipids in the blood. Your participation is voluntary. You have the right to refuse to participate in this study. If you decide to participate, you may still choose to withdraw from the study at any time without any negative consequences to the medical care, education, or other services to which you are entitled or are presently receiving. All information collected about you during your participation in the study will be kept for analysis. Before you decide, it is important for you to understand what the research involves. The consent form will tell you about the study, why the research is being done, what will happen to you during the study and the possible benefits, risks and discomforts. You also need to know that there are important differences between being in a research study and being cared for by your doctor. When you participate in a research study, the main goal is to learn things to help other patients in the future. Outside a research study, your doctor’s only goal is to care for your health. Nevertheless, the researchers have a duty of care to all subjects and will inform you of any information that may affect your willingness to remain in the study. If you wish to participate in this study, you will be asked to sign the consent form. Please take time to read it carefully and to discuss it with your family, friends, and doctor before you decide.
Confidentiality
Your confidentiality will be respected according to the Canadian Personal Information Protection and Electronic Documents Act (PIPEDA S.C.2000c.5). No information or records that disclose your identity will be published without your consent, nor will any information or records that disclose your identity be removed or released without your consent unless required by law. You will be assigned a unique study number as a subject in this study. Only this number will be used on any research related information collected about you during the course of this study, so that your identity [i.e. your name or any other information that could identify you] as a subject in this study will be kept confidential. Your rights to privacy are legally protected by federal and provincial laws that require safeguards to insure that your privacy is respected and also give you the right of access to the information about you that has been provided to the sponsor and, if need be, an opportunity to correct any errors in this information. If you choose to enter the study and decide to withdraw, which you may do at any time, all data collected about you during your enrollment in the study will be retained for analysis but will be kept anonymous. To withdraw from this study, simply notify the study coordinator (listed on your signed consent form) in writing.
How to participate?
- Make sure you meet the requirements to participate to this study (see “Who can participate?” section above).
- Talk to your general practitioner or family doctor about your involvement in the FH Canada Registry, he or she may want to access the “Health Care Professionals” section for details and/or become a FH Canada participating clinician.
Who to Contact
If you think you have FH and would like to register in the FH Canada registry, please find a specialized lipid clinic near you. If you need additional assistance or have any question about the FH Canada Registry, please contact our National Coordinator.
Contact
Isabelle Ruel, PhD
FH Canada National Coordinator
Research Institute of the McGill University Health Centre
1001 Decarie Boul. E01.3144
Montreal, QC
Canada H4A 3J1
Phone: 514-934-1934 ext 34852
Fax: 514-933-6418
[email protected]
Additional Resources
Educational resources
- Video: FH Canada - What is Familial Hypercholesterolemia?
- The History of Familial Hypercholesterolemia
- Presentation on the FH Canada Registry
- Familial Hypercholesterolemia for patients
- Learning About Familial Hypercholesterolemia
- Patient’s viewpoint on FH published in Circulation Cardiovascular Quality and Outcomes
- Familial Hypercholesterolemia; National Organization for Rare Disorders
- Learn your lipids
-
iCardio website

Info on drugs
- Alirocumab pregnancy exposure registry
- Evolocumab pregnancy exposure registry overview
- Evolocumab pregnancy exposure registry flyer
- PCSK9: Repatha – Evolocumab Common Drug Review document
- Evolocumab (trade name Repatha), exceptional medication for HeFH in Quebec
- PCSK9: Praluent – Alirocumab Common Drug Review document